You are here
Foreign Components in eRPM
When working with foreign components in eRPM, you'll see question 5.13 on the PAF. This relates to Foreign Components in research or the portion of project work that will be performed outside the United States.
 PAF Question 5.13- Significant Effort Outside of United States - FAQs
PAF Question 5.13- Significant Effort Outside of United States - FAQs
The eResearch Proposal Management (eRPM) system Proposal Approval Form (PAF) has a required question 5.13 that relates to the foreign component of work, or the portion of project work that will be performed outside the United States. If the answer is yes, you are asked to select the country(ies) from a drop down list.
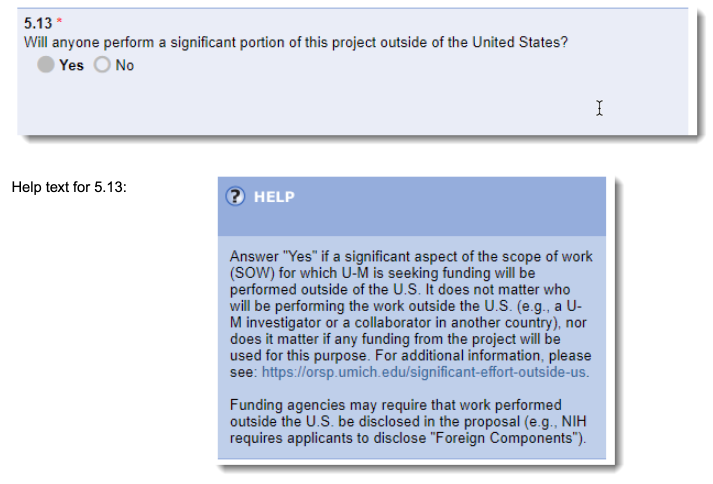
eRPM Question 5.13:
"Will anyone perform a significant portion of this project outside of the United States?"
Help Text: "Yes" if a significant aspect of the scope of work (SOW) for which U-M is seeking funding will be performed outside of the U.S. It does not matter who will be performing the work outside the U.S. (e.g., a U-M investigator or a collaborator in another country). Nor does it matter who is funding the work, if at all. Funding agencies may require that work performed outside the U.S. be disclosed in the proposal (e.g., NIH requires applicants to disclose “Foreign Components”).
Additional U-M FAQs to Aid in Answering Question 5.13
FAQs
Yes. There are three potential reporting requirements in this scenario:
- Foreign Component: For proposals and progress reports, you will need to account for this visitor as a “Foreign Component” if (a) s/he is performing a significant element of the project and (b) s/he is performing at least some portion of the project outside of the United States.
- Other Support: For proposals and progress reports, the visitor should be disclosed as a source of non-monetary Other Support.
- Project Personnel: For progress reports, the visitor should be identified as a participant if s/he has worked at least one person month per year on the project during the reporting period, regardless of funding source.
Yes. You may not perform a significant scientific element or segment of an NIH-funded project outside the U.S. without prior NIH approval. Per NIH Grants Policy Statement 8.1.2.10 “Adding a foreign component under a grant to a domestic or foreign organization requires NIH prior approval.”
The NIH definition of “foreign component” states that “extensive foreign travel by recipient project staff for the purpose of data collection, surveying, sampling, and similar activities” is a “foreign component.”
Please reach out to your NIH Grants Management Specialist or Program Officer to confirm whether or not these individuals need to be listed. When required, disclosure of foreign co-authors to the NIH should occur prior to publication. Other sponsors have not specifically commented on this. Should you have questions, please consult your ORSP Project Representative.
Just because your sponsor is a foreign entity doesn’t mean your project has a foreign component. It’s where the work is performed that determines whether there’s a foreign component.
No, this is not performance of the project.
This is not performance of a portion of the project and you should answer "no" unless the work is performed in a foreign country.
The recipient institution should evaluate the element of the project that is being conducted outside of the United States within the context of the project as a whole when making determinations about significance. Some examples of activities that may be considered a significant element of the project include, but are not limited to:
- collaborations with investigators at a foreign site anticipated to result in co-authorship
- use of facilities or instrumentation at a foreign site
- receipt of financial support or resources from a foreign entity
Same as above. (See answer to “What if the sponsor is a foreign entity.”)
You are completing the PAF for U-M’s portion of the project.
Per NIH guidance, For post-docs and graduate students that are required to work on their originally approved work remotely from a foreign country due to COVID-19 travel restrictions, where no grant funds are going to a foreign entity, NIH has determined that this scenario does not constitute the performance of a significant scientific element or segment of the project outside the US, as outlined in the NIH Grants Policy Statement definition of a foreign component.
References and Resources
-
SOP 200.05 Foreign Component Policy
November 30, 2020
Questions?
- If you have questions (that are not related to your school’s or college’s implementation of this policy), reach out to your ORSP project representative.