You are here
What is an office of research and sponsored projects?
Let's start with an understanding of a research institution. In addition to teaching students and administering degrees, research is integral to the pursuit of scholarship. Research universities, like the University of Michigan, conduct research as a core part of their mission.
Students, research project teams, benefit from the research process, and all, including the public, benefit from the discoveries and outcomes.
The federal government mandates that a central unit at universities administer research activities funded by sponsors.
As a public institution, the University of Michigan's mission is to generate and disseminate knowledge in the public interest for open scholarly exchange and academic freedom. These principles are referred to as "Openness in Research" and are set forth by the Regents' policy, under the Standard Practice Guide (SPG) 303.02.
A university's research and sponsored projects works with both faculty and external sponsors to manage and ensure compliance with proposals, awards, contracts and agreements, subrecipients, closeouts and more.
What is a sponsored project?
In the most simple terms, a sponsored project is a grant or an award funded by an external sponsor.
When universities conduct sponsored research, that research is funded by an external sponsor. For example a sponsored project may be funded by a government agency or by a private sponsor, such as a corporation or a non-profit organization.
Faculty who conduct research (investigators) write grants to sponsors to fund the research.
What is an external sponsor?
An external sponsor invests in research to advance knowledge and discovery.
These sponsors might be government (federal agencies, states, or municipalities).
The largest source of external sponsored funding typically comes from government agencies (the National Institutes of Health, the National Science Foundation, the Department of Defense, Agriculture, Energy or more).
Or the sponsor might be private, as a non-profit or foundation (such as the American Heart Association) or as a for-profit company or industry. The grants help cover the equipment, facilities, and staff for the research activities.
These sponsors may issue a grant, contract, or cooperative agreement to the university. When an award is granted by a sponsor, it's time to set up the project and begin the research, managing the project through its completion or close out.
What is research administration?
The field of research administration involves vigilance and a broad understanding of:
- Policy and agreements with sponsors (whether through contracts, terms and conditions, university policy, or federal regulations)
- allowable and unallowable costs
- Knowledge of the types of third party agreements that may be required
- Proposal budget preparation and how to apply the institution's F&A cost rate;
- Assist a Principal Investigator in finding sources of funding (research development)
- The requirements before a project is awarded.
- Financial reporting
- Non-financial milestone reporting
- Publication and data sharing responsibilities
- Closeout of a sponsored project and audit preparation.
- Compliance areas, including human subjects protections, animal care, export controls, data sharing, research misconduct, subrecipient monitoring, effort reporting, controlled substances, and more.
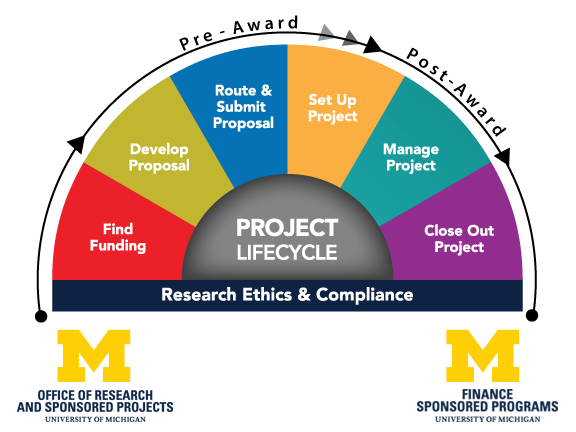
Understanding the Research Project Lifecycle
Our processes and our website are organized according to the lifecycle of a research project to enable understanding of sponsored projects from the beginning phases (pre-award) to the award start and completion phases (post-award). Learn more on our Getting Started page.
Contact [email protected] with questions!
FAQs
Funding searches are handled by the schools/colleges at the unit level. Grants and funding requests are not written and submitted independently. The federal government mandates that a central unit at universities administer research activities funded by sponsors.
These administrative tasks require a professional staff with broad knowledge of the regulations of federal agencies and to maintain the extensive records required by federal sponsors. Start by working with your Research Administrator.
The Office of Research and Sponsored Projects (ORSP) primarily handles pre-award activities and is part of the Office of Research.
Sponsored Programs primarily handles financial post-award activities, and is part of Finance.
In eRPM:
- For projects that are not a clinical trial, you need only select "Private Sponsor Team" in eRPM from the list of Project Representatives. When you do, ORSP will take on the task of determining the PR.
- For projects that are clinical trials and originate in the Cancer Center, Internal Medicine and Pediatrics, select Lark Haunert for sponsors "A" through "L" or select Tricia Haynes for sponsors "M" through "Z."
- For clinical trials that originate in other departments (other than the Cancer Center, Internal Medicine and Pediatrics) select Debra Dill.
For all other questions:
- Contact Reynaldo Martell ([email protected], 936-1353), the Administrative Specialist for the Private Sponsors Team. Just share the name of your project's Principal Investigator (PI), your Department or School, and Sponsor, and they can assist you and connect you to the right PR.
So that we can best serve you, when contacting us, please include:
- Proposal Approval Form (PAF) number
- Project Grant (P/G)
- UFA (Unfunded Agreement) number
- SSP (Special Sponsored Project) number
- PI (Principal Investigator) name
- Sponsor name and detailed contact information (if requesting that a central office contact them)
References and Resources
- Navigating the ORSP Website - Wayfinding Video Tutorials
- Navigate Overview of the Research Project Lifecycle
-
ORSP - Useful Web Resources
Useful Web Resources for Research Administrators - Annotated by ORSP Communications. Organized by the Research Project Lifecycle.
- Frequently Required Proposal Documents and Data